
Aug 3, 2022Interpreting your pecan leaf sample results
I have written previously on the importance of taking foliar samples of your pecan orchard to determine nutrient status. This year, let’s look at an example of a foliar sample report and make recommendations based on the results. Ideally you have a soil report in hand, as well, to make better informed decisions. In this case we do not, but I know from past experience that this orchard is in an alkaline soil type. To make the best decisions for your orchard make sure you know your soil pH at the very least. If you fertilize blindly without it, you may be literally throwing money away or even damaging your trees.
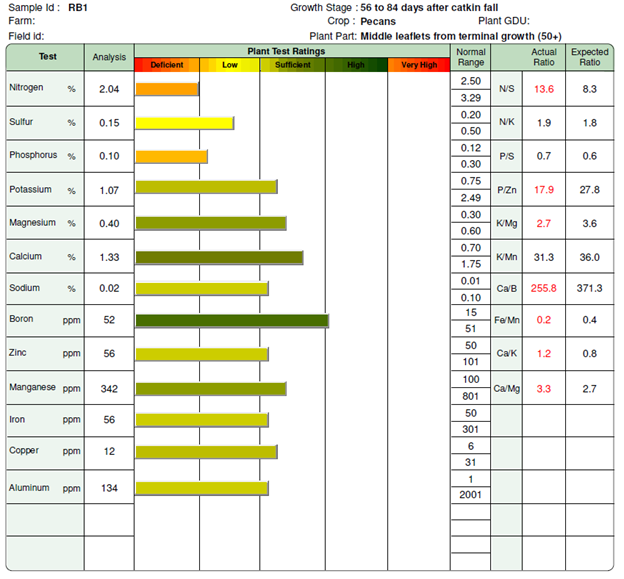

When I look at this foliar test, the first things that stand out are that nitrogen is deficient, while phosphorus and sulfur are low. Everything else is sufficient or high. Just because something is sufficient does not mean you should not take a closer look at it. Though the iron and zinc (Zn) levels are sufficient in this test, they are on the lower level, and I would strongly consider putting one out this year. Since we know that zinc is critical for pecan production, I would recommend an application while we are on the lower sufficient level rather than waiting for it to become deficient. I think we can probably wait on the iron, but keep it in mind for next year.
Let’s start with those that are deficient and low: nitrogen (N), phosphorus (P) and sulfur (S). Of these, I am going to single out S first. Sulfur applications vary with your soil pH. For an acidic soil, you are going to want to be careful since it will make your soil even more acidic. In some cases, it may even need to be paired with a lime application to ensure you are not bringing your soil to dangerously acidic levels. For this particular orchard, I expect the pH to be alkaline, so growing conditions will likely improve from a slight acidification. I generally recommend that sulfur be added to orchards in the form of ammonium sulfate so that you can get your nitrogen at the same time. Occasionally, it can be worthwhile to consider an application of zinc sulphate to get some zinc instead, but it tends to be more expensive than ammonium sulphate.
In the comments section of this particular test, it recommends 20 to 50 lbs./ac. of nitrogen. Deciding how much to put out in this situation depends on your personal production goals and orchard growing conditions.
I usually favor lower to medium rates of N application. When considering lower application rates, it is sometimes worthwhile to consider two split applications which cumulatively bring you to a higher rate. Since we are low on our sulfur as well, I would strongly consider putting out ammonium sulphate (21% N and 24% S). With its composition, a rate of 100 lbs./ac. would put 21 lbs. of N and 24 lbs. of S per acre.
The comments for sulfur on the test were in the 20 to 30 lbs./ac. range, so we would fulfill both recommendations at the same time. Depending on the state of the orchard, it might be worthwhile to consider additional N, but with fertilizer prices high as they are, I believe that a low application should be adequate for this particular one.
We don’t want to forget about the phosphorus either. A good N to P ratio helps ensure good growth and prevent leaf burn. The comments specify 30 to 40 lbs. of P2O5 per acre. Make sure to read your reports carefully since some will report it in lbs. of P instead, which can require an extra conversion to determine application rates (P2O5 is only 43.7% P for those that need to do the extra step). Phosphorus binds with soil exchange sites easily and can move slowly in the soil. Some research has indicated that you can apply P in a band to make it more readily available to the tree.
For this orchard, I would recommend either a banded or broadcast application of triple superphosphate (0-46-0) depending on the producer’s preference. Triple superphosphate is 46% P2O5. This translates to ~65 to 85 lbs./ac. to reach the needed P levels. If I were going with a band application, the 65 lbs./ac. should be sufficient. For a broadcast application, I would consider the higher rate of 85 lbs./ac.
Revisiting our zinc, we have several options at our disposal. One of them is doing nothing since we currently have sufficient levels. As mentioned earlier, they are on the lower side of sufficiency, so I would consider an application of some sort to keep them within sufficient levels. Many growers favor adding Zn in their spray applications – if this is you, then stay the course.
I have had several inquiries about soil-based applications. There is research that indicates that it is effective, but that it can take a while to be effective. With that in mind, the time to make an application is when you are still in sufficient levels rather than waiting for deficiency. This is a situation where I really wish I had a soil test to see what is there already. Another factor to consider is that Zn becomes less available to tree roots at an alkaline pH, which I suspect this orchard has. Going in blind, I would say we could safely add at least 5 lbs. of Zn per acre and hope that the S we are adding helps with the availability.
The most common form of Zn available is zinc sulfate (35% Zn and 16.5% S). To get our 5 lbs. of Zn per acre we will need to put out ~14 lbs. of zinc sulfate. In a different orchard with a more acidic pH, I might be mildly concerned about the S that would be added, but in this orchard it might be beneficial instead.
If you need help making sense of your soil and foliar tests, feel free to contact me. You can email me at MPolozola@agcenter.lsu.edu, or give me a call at my office at 318-427-2669.
– Michael Polozola, Lousiana State University







